Demystifying Photobleaching Technology in Photoinitiators
In fields like UV-curable coatings, 3D printing, and dental fillings, a key technology known as "photobleaching" operates behind the scenes. It doesn't refer to the traditional removal of color. Instead, it describes a process where specific photoinitiators absorb light energy and then actively destroy their own light-absorbing structure, transforming from colored to colorless. This "self-sacrifice" offers two major benefits: it allows light to penetrate deeper, enabling thorough curing of the material; and it eliminates the initiator's own color, resulting in a clear, transparent final product. This article will delve into this fascinating chemical reaction occurring in the microscopic world and explore how it shapes many high-tech products in our lives.
1. What is Photobleaching?
Photobleaching is a unique property characteristic of certain photoinitiators.
Under exposure to ultraviolet or visible light, it first absorbs the light energy. Then, it essentially "breaks apart" – undergoing a permanent chemical structural change. It transforms from a colored substance capable of absorbing visible light into a colorless substance that no longer absorbs light. Macroscopically, we observe the material changing from a faint yellow hue to colorless and transparent, as if it has been "bleached."
2. The Fascinating Principle of Photobleaching: The "Suicidal" Destruction of Chromophores
Photoinitiators exhibit color (typically yellow) because their molecules contain "chromophores" – groups capable of absorbing light of specific wavelengths.
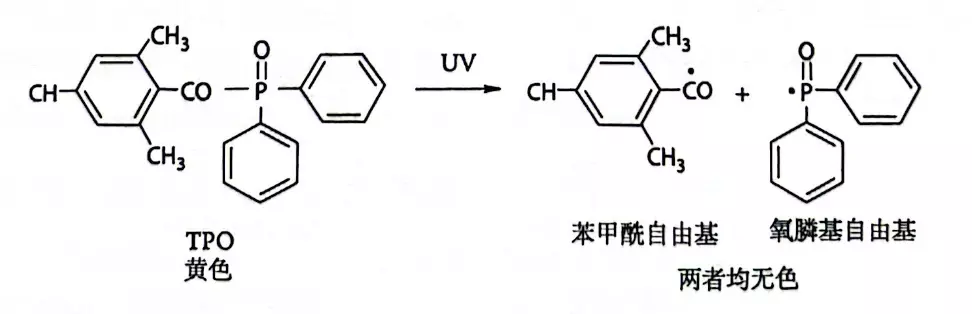
For conventional photoinitiators, their chromophore structure may still exist after the reaction, causing the final product to exhibit a slight yellow tint. In contrast, photobleaching photoinitiators (most commonly cleavage-type/Type I) take a more decisive approach.
Taking the star product, acylphosphine oxide (such as TPO), as an example:
1.Light Absorption: It absorbs energy in the ultraviolet (UV) region.
2.Bond Cleavage: A critical chemical bond within the molecule breaks, fragmenting it into two highly reactive free radical species.
3.Chromophore Destruction: The molecular structures of these newly generated fragments are completely different from the parent molecule. The original chromophore is utterly destroyed.
4.Becoming Colorless: The new fragments cease to absorb visible light above 400 nm (in the violet-blue spectrum). Consequently, the yellow color of the material disappears, resulting in a transparent appearance.
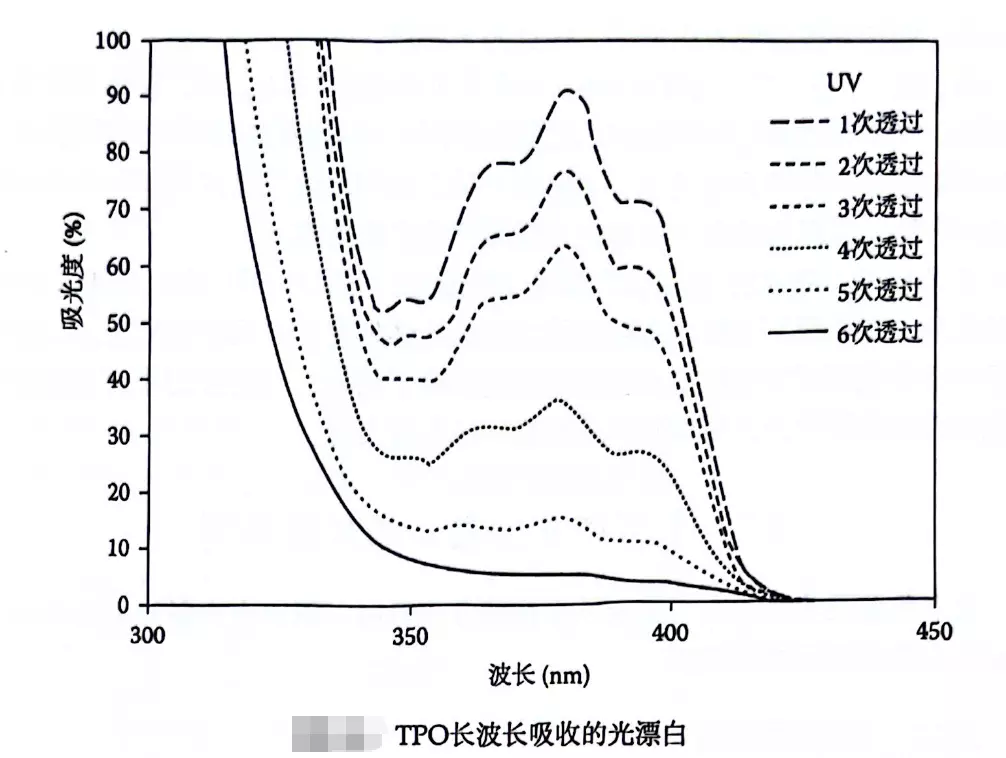
This process is akin to a cup of strong tea. Under light exposure, the colored substances within it decompose, causing the tea to gradually become clear from top to bottom.
3. The Two Core Advantages Brought by Photobleaching
This "self-sacrificing" technology delivers two crucial benefits:
1. Achieving Deep Cure, Eliminating "Superficial Work":
Without photobleaching, the photoinitiator acts like a layer of "sunscreen," strongly absorbing and blocking the light. The result is a hard, cured surface with the underlying layers remaining sticky. The photobleaching effect removes these "roadblocks," allowing the light to penetrate deeply and cure the entire coating uniformly. Even thick layers of clear varnish or fillers can be cured thoroughly.
2. Obtaining Colorless Products, Achieving Ultimate Aesthetics:
Many high-end applications, such as coatings for mobile phone cases, clear topcoats, or dental filling materials, have extremely stringent color requirements. Photobleaching initiators essentially "disappear" after completing their task. This prevents the product from yellowing due to residual initiator, ensuring the final product is crystal clear and possesses pure coloration.
4. The Stars and Application Stages of Photobleaching Technology
Star Molecules:
Acylphosphine Oxides (TPO, Irgacure 819): These are currently among the most widely used and effective photobleaching initiators. They are extensively employed in wood coatings, plastic coatings, and 3D printing resins to achieve deep cure and high transparency.
Camphorquinone (CQ): This initiator absorbs blue light and appears yellow. It is primarily used in dental composites. Although it has a slower curing speed, its excellent photobleaching effect allows the filling material to present a very aesthetically pleasing white, perfectly meeting the demands of dental aesthetics.
Conclusion
Photobleaching, this seemingly subtle chemical property, is actually the unsung hero driving the development of numerous cutting-edge manufacturing technologies. It perfectly embodies the unity of "function and form" – through molecular-level "self-sacrifice," it not only efficiently fulfills the mission of curing but also achieves the ultimate performance and beauty of the product. Next time you admire a smooth, transparent UV-coated product or use a high-precision 3D printed part, you might recall this "transparent magic" called photobleaching happening in the microscopic world.





